Hepatitis
B Treatment Improves Liver Disease in HIV/HBV Coinfected
 |
 |
 |
 |
| SUMMARY:
Dual-acting nucleoside/nucleotide analogs
including tenofovir (Viread) suppressed
HBV viral load and slowed liver disease
progression in HIV positive patients in
Spanish study. |
|
 |
 |
 |
 |
By
Liz Highleyman
Due to overlapping transmission methods, many people
with HIV are also
exposed to hepatitis B
virus (HBV), and some develop chronic HIV/HBV
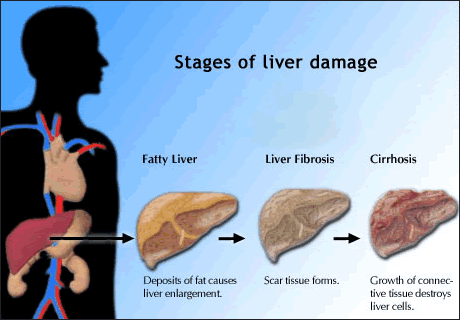
coinfection. Over time, hepatitis B can lead to
advanced liver fibrosis, cirrhosis, and hepatocellular
carcinoma (liver cancer). On average, HIV/HBV coinfected
individuals experience more rapid liver disease progression
than people with HBV alone.
Treatment
guidelines recommend that coinfected patients should
receive antiretroviral
therapy (ART) regimens that contain drugs with
dual activity against both HIV and HBV, which include
tenofovir
(Viread, also in the Truvada
and Atripla
coformulations), lamivudine
(3TC, Epivir), and emtricitabine
(Emtriva).
As
described in the January
2, 2011, issue of AIDS, Luz Martin-Carbonero
from Hospital Carlos III in Madrid and colleagues
evaluated the long-term effects of dually active nucleoside/nucleotide
analogs in a cohort of coinfected patients in Spain.
Over
a median follow-up period of 35 months, the researchers
retrospectively analyzed changes in HBV DNA viral
load, hepatitis B "e" antigen (HBeAg) and
hepatitis B surface antigen (HBsAg), and clinical
outcomes including fibrosis progression (estimated
using the non-invasive FibroScan method), hepatocellular
carcinoma, decompensation or liver failure, and death.
The analysis included 92 patients with HIV/HBV coinfection;
about 30% also had hepatitis C virus (HCV) and about
20% were also infected with hepatitis delta virus
(HDV). Most (88%) were men, the median age was 41
years, and 45% were initially HBeAg positive. At baseline,
48% had absent or mild liver fibrosis, 28% had moderate-to-advanced
fibrosis, and 24% had cirrhosis. Overall, 94% received
ART containing lamivudine or emtricitabine, and 82%
used tenofovir.
Results
 |
13
patients were lost to follow-up, leaving 79
included in the analysis. |
 |
71
of 79 participants, or 89%, achieved undetectable
HBV DNA. |
 |
11
of the 42 HBeAg positive patients cleared HBeAg
(9.0 per 100 person-years), and 5 also experienced
anti-HBe antibody seroconversion. |
 |
7
participants experienced HBsAg clearance (2.6
per 100 person-years), 4 of whom also had anti-HBs
antibody seroconversion; 2 people in this group
were triply infected with HDV. |
 |
During
follow-up 8 patients -- half of whom also had
HDV -- progressed to liver decompensation (2.9
per 100 person-years) and 1 developed hepatocellular
carcinoma. |
 |
6
patients died (2.2 per 100 person-years), with
4 deaths attributable to liver disease. |
 |
Among
71 patients who received follow-up FibroScan
evaluations after a median of 40 months: |
 |
 |
75%
showed no changes in fibrosis stage; |
 |
17%
experienced improvement; |
 |
17%
showed worsening fibrosis. |
|
 |
HCV
triple infection and low CD4 T-cell count were
significant risk factors for liver cirrhosis. |
Based
on these findings, the study authors concluded,
"Most HIV/HBV-coinfected patients treated with
anti-HBV active [nucleoside/nucleotide analogs]
experience an amelioration of liver fibrosis progression,
with low rates of hepatic decompensation and death."
"Serum HBeAg or HBsAg seroconversion occurs
at yearly rates of 9[%] and 2.6%, respectively,
even in patients with delta hepatitis," they
added.
Nevertheless, they noted, "oral [nucleoside/nucleotide
analogs] do not entirely annul the risk of hepatic
decompensation events or death in this population,
including the development of hepatocellular carcinoma."
Investigator affiliation: Department of Infectious
Diseases, Hospital Carlos III, Madrid, Spain.
3/29/11
Reference
L Martin-Carbonero, T Teixeira, E Poveda, et al.
Clinical and virological outcomes in HIV-infected
patients with chronic hepatitis B on long-term nucleos(t)ide
analogues. AIDS 25(1): 73-79 (abstract).
January 2, 2011.